Gangrene-causing bacteria could treat late-stage cancers – study
Conducted by doctors at the University of Texas, the ground-breaking study saw late-stage cancer patients’ tumours injected with spores of a modified bacterial strain related to the lethal hospital superbug, clostridium difficile.
The first phase of the study involved 24 patients with solid tumours that had become resistant to existing treatments such as chemotherapy.
Known as Clostridium novyi-NT, the strain thrives in the dense cancerous tissues as it doesn’t need high quantities of oxygen to survive. Healthy tissue meanwhile, is rich in oxygen so the bacteria won’t develop.
Two patients who were given the highest doses developed severe sepsis and “gas gangrene.” But, for the remaining 22 patients, this new type of “bacterial therapy” instead attacked the cancerous tissues causing them to become necrotic and shrink.
“Even after a single injection of this bacterial therapy, we see biological and, in some patients, clinically meaningful activity,” said Dr Filip Janku of the university’s Anderson Cancer Center ahead of the study being presented at the International Cancer Immunotherapy Conference in New York on Sunday.
READ MORE: Cancer mapping tech could help UK doctors stay one step ahead of tumors
In 23 percent of cases, the tumours shrank in size by more than 10 per cent and Janku is confident that even greater shrinkage was possible as the initial treatment caused inflammation of the tumours while they were being attacked.
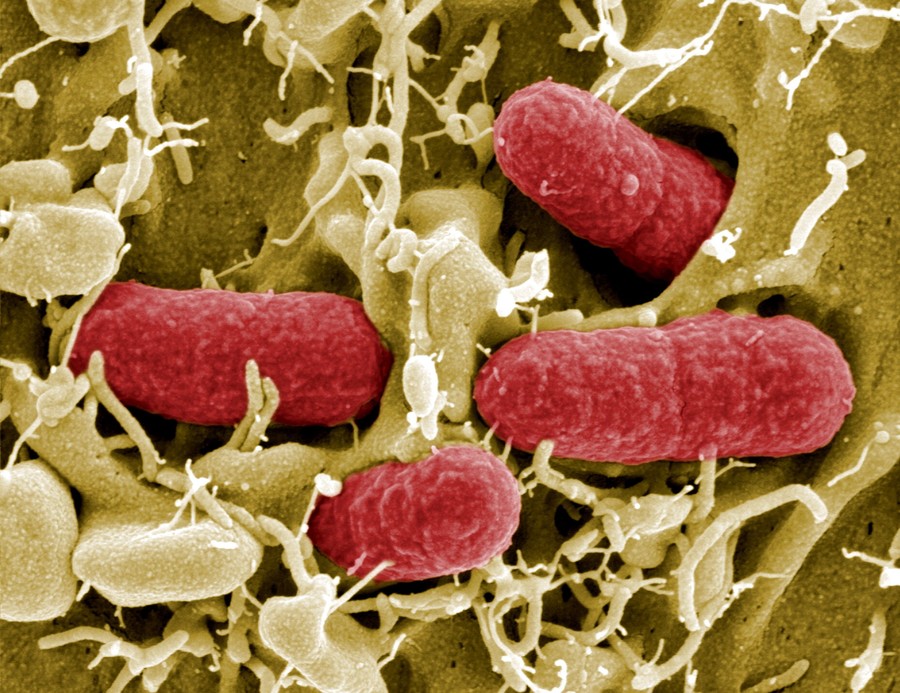
“Extremely encouraged” by the results of this trial, Janku added that even when the spores did not develop, the presence of the bacteria helped to active dormant immune system cells to also attack the tumour.
While the treatment has yet to be published in a scientific journal or go through the significant process of human testing for safety and long-term trails, Janku believes this “bacteriolytic strategy” could be “clinically meaningful” when used with existing immunotherapies.













Add new comment